In eukaryotes, a preinitiation complex forms made of the small 40S subunit, the initiator Met-tRNAi, and eIF2-GTP. In archaea, translation initiation is similar to that seen in eukaryotes, except that the initiation factors involved are called aIFs (archaeal inititiaion factors), not eIFs. The remaining eIFs dissociate from the ribosome and translation is ready to begins. The 60S ribosomal subunit is recruited to the pre-initiation complex by eIF5-GTP, which hydrolyzes its GTP to GDP to power the assembly of the full ribosome at the translation start site with the Met-tRNAi positioned in the ribosome P site. After this, eIF2-GDP is released from the complex, and eIF5-GTP binds. Once the appropriate AUG is identified, eIF2 hydrolyzes GTP to GDP and powers the delivery of the tRNA i-Met to the start codon, where the tRNA i anticodon basepairs to the AUG codon. The nucleotides around the AUG indicate whether it is the correct start codon. Many, but not all, eukaryotic mRNAs are translated from the first AUG sequence. Once at the cap, the pre-initiation complex tracks along the mRNA in the 5' to 3' direction, searching for the AUG start codon. Poly (A)-Binding Protein (PAB) binds both the poly (A) tail of the mRNA and the complex of proteins at the cap and also assists in the process. Several other eIFs, specifically eIF1, eIF3, and eIF4, act as cap-binding proteins and assist the recruitment of the pre-initiation complex to the 5' cap. The eukaryotic pre-initiation complex then recognizes the 7-methylguanosine cap at the 5' end of a mRNA. The initiator tRNA, called Met-tRNA i, carries unmodified methionine in eukaryotes, not fMet, but it is distinct from other cellular Met-tRNAs in that it can bind eIFs and it can bind at the ribosome P site. In eukaryotes, a pre-initiation complex forms when an initiation factor called eIF2 ( eukaryotic initiation factor 2) binds GTP, and the GTP-eIF2 recruits the eukaryotic initiator tRNA to the 40s small ribosomal subunit. This interaction anchors the 30S ribosomal subunit at the correct location on the mRNA template. coli mRNA, a sequence upstream of the first AUG codon, called the Shine-Dalgarno sequence (AGGAGG), interacts with the rRNA molecules that compose the ribosome. When an in-frame AUG is encountered during translation elongation, a non-formylated methionine is inserted by a regular Met-tRNA. coli, and is usually clipped off after translation is complete. Formylated methionine is inserted by fMet-tRNA at the beginning of every polypeptide chain synthesized by E. Methionine is one of the 21 amino acids used in protein synthesis formylated methionine is a methione to which a formyl group (a one-carbon aldehyde) has been covalently attached at the amino nitrogen. The initiator tRNA basepairs to the start codon AUG (or rarely, GUG) and is covalently linked to a formylated methionine called fMet. 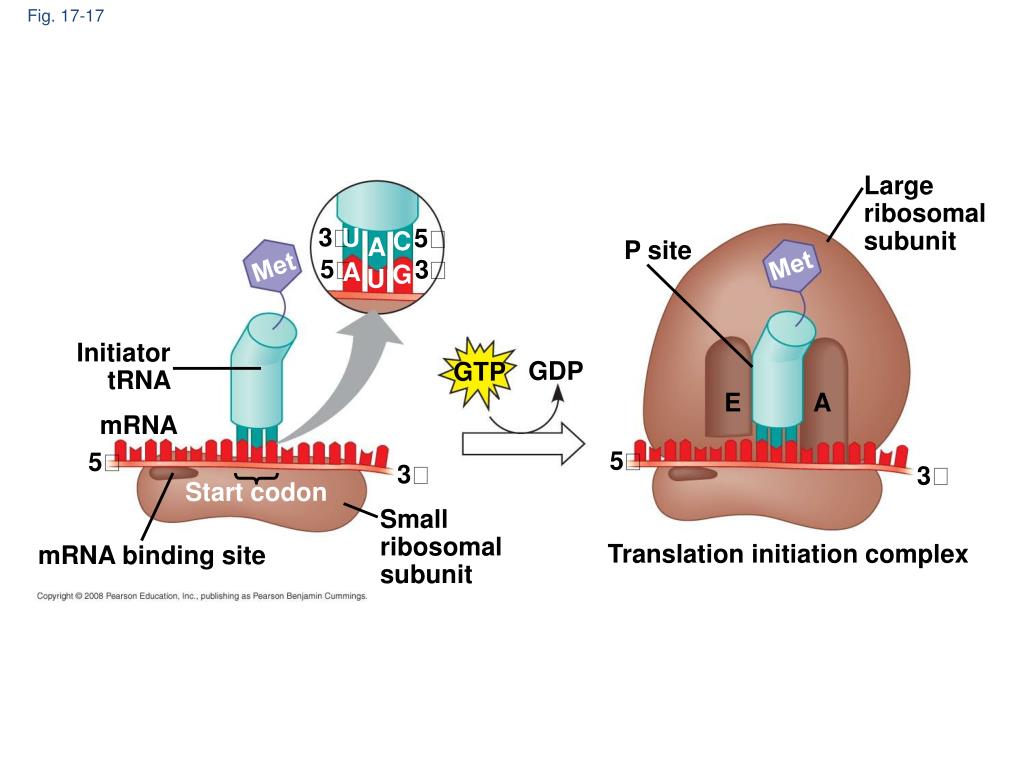 
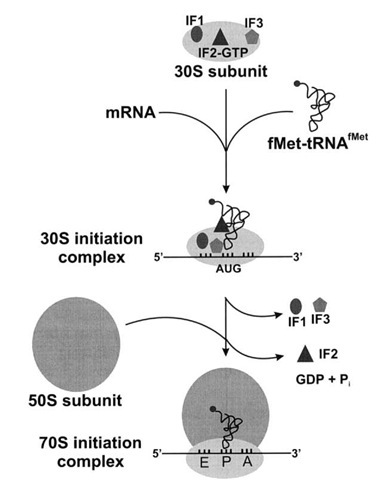
coli, this complex involves the small 30S ribosome, the mRNA template, three initiation factors (IFs IF-1, IF-2, and IF-3), and a special initiator tRNA, called fMet-tRNA. Protein synthesis begins with the formation of a pre-initiation complex. The charged initiator tRNA, called Met-tRNA i, does not bind fMet in eukaryotes, but is distinct from other Met-tRNAs in that it can bind IFs.As with mRNA synthesis, protein synthesis can be divided into three phases: initiation, elongation, and termination. In eukaryotes, a similar initiation complex forms, comprising mRNA, the 40S small ribosomal subunit, IFs, and nucleoside triphosphates (GTP and ATP). Guanosine triphosphate (GTP), which is a purine nucleotide triphosphate, acts as an energy source during translation-both at the start of elongation and during the ribosome’s translocation. When an in-frame AUG is encountered during translation elongation, a non-formylated methionine is inserted by a regular Met-tRNA Met. coli, but it is usually clipped off after translation is complete. \) at the beginning of every polypeptide chain synthesized by E.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
